
APHL and FDA Partner on Cronobacter Detection Training
In September 2025, APHL partnered with the US Food and Drug Administration’s (FDA’s) Office of Training, Education and Development (OTED) and the Office of Laboratory Operations and Applied Science (OLOAS) within the Human Foods Program (HFP) to deliver a first of its kind, in-person laboratory training on detecting Cronobacter spp. in powdered infant formula (PIF). Cronobacter spp. is an emerging opportunistic pathogen capable of surviving in dry environments such as PIF and causing rare, severe and sometimes fatal illness in neonates and infants. Through multidisciplinary collaboration, FD430 Cronobacter spp. Laboratory Training: Detection in Powdered Infant Formula translated a complex, technical topic into practical, hands-on learning. While APHL has collaborated with FDA on training initiatives for more than a decade, this effort marked the first time APHL developed and delivered an FDA in-person course from concept through execution.
A Critical Training Need
Several events elevated this training to a top priority. In 2022, an investigation of Cronobacter sakazakii linked to PIF from a major manufacturer resulted in serious illnesses, a nationwide recall and significant formula shortages— revealed critical gaps in state and federal laboratory testing capacity. In response, FDA and key collaborators launched a Cronobacter Prevention Strategy and prioritized strengthening testing capabilities through standardized methods and targeted training. As improved testing procedures were developed, the need for comprehensive training across federal and state laboratories became urgent, positioning the FD430 Cronobacter spp. Laboratory Training as a high FDA priority.
Creators Behind the Course
The success of the course reflects extensive collaboration among FDA, APHL and state laboratories. The FD430 Course Advisory Group (CAG) included leading FDA microbiologists at the forefront of Cronobacter testing. Among them were the current authors of the FDA Bacteriological Analytical Manual (BAM) Chapter 29—Cronobacter, including Yi Chen, Nancy E. Miranda, Kun C. Liu and Jeremi S. Mullins.
In addition to these subject matter experts, FDA provided essential training coordination and project management support, while APHL contributed expertise in on-site logistics and instructional design. Guided by the BAM and the expertise of the CAG, APHL created materials grounded in cognitive science and adult learning principles to improve engagement and retention.
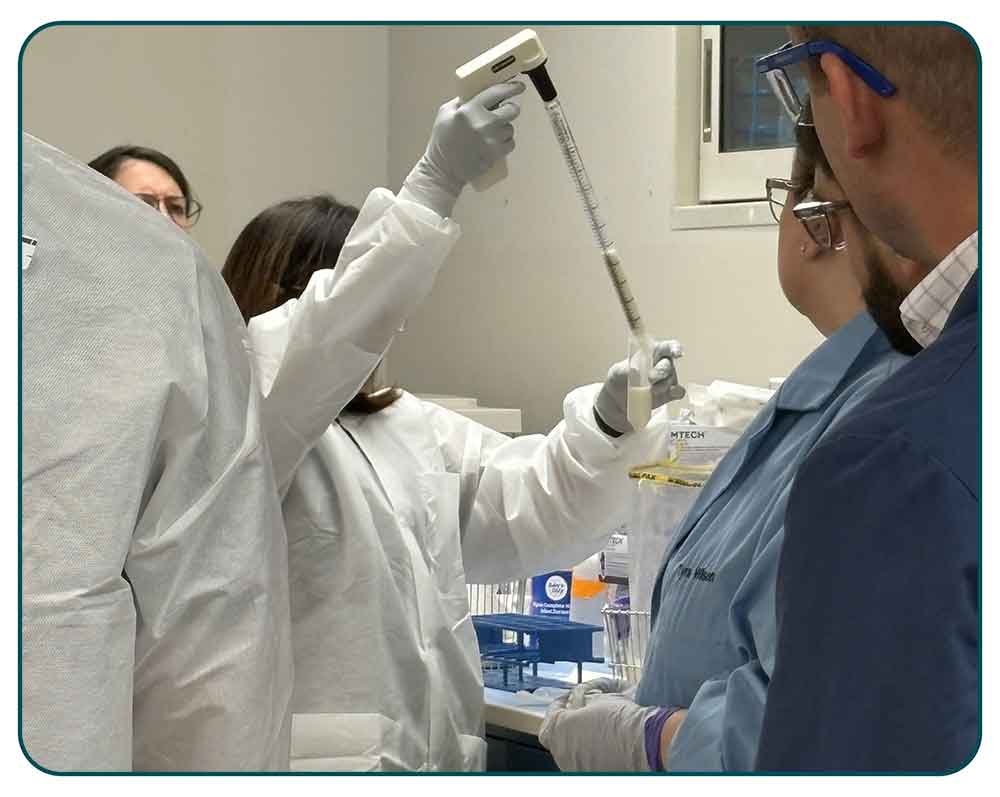
An instructor demonstrates how to remove supernatants from a powdered infant formula enrichment.
The CAG evaluated delivery modality (in-person, virtual or online), training location, target participants and engagement strategies to ensure the course addressed real-world laboratory needs. This multi-organizational, cross-functional collaboration paired FDA’s scientific leadership with APHL’s learning design expertise to create a course that was both technically rigorous and learner-centered.
Training Delivery
Hosted at FDA’s Seattle Human and Animal Food Laboratory (SEAHAF), the training combined classroom instruction with hands-on laboratory practice. The 14 trainees represented both state and FDA food laboratories. Blending technical instruction with real-time practice and discussion proved invaluable. Learners exchanged insights with experts, built connections between state and federal partners and learned directly from one another.
Participants praised the course as well-planned and highly relevant, highlighting the usefulness of interactive materials such as the participant workbook and the expertise of FDA and APHL staff. The knowledge gained will enable attendees to train others in their laboratories, helping address critical gaps in state and federal laboratory capacity to detect Cronobacter in PIF. This training demonstrated how blending scientific expertise with instructional design—and engaging teams across organizations—can produce learner-centered programs driven by a shared mission to protect public health.

Participants, instructors and APHL staff outside of FDA’s Seattle Human and Animal Food Laboratory in Bothell, WA.
Future Training Initiatives
FD430 reinforced the urgency of strengthening Cronobacter testing for food safety, particularly for vulnerable populations. Beyond technical skills, the course showcased the power of partnership: APHL, FDA and state agencies aligning science and learning design toward a common goal. APHL intends to apply this collaborative approach to future training initiatives that advance food safety testing capabilities.

