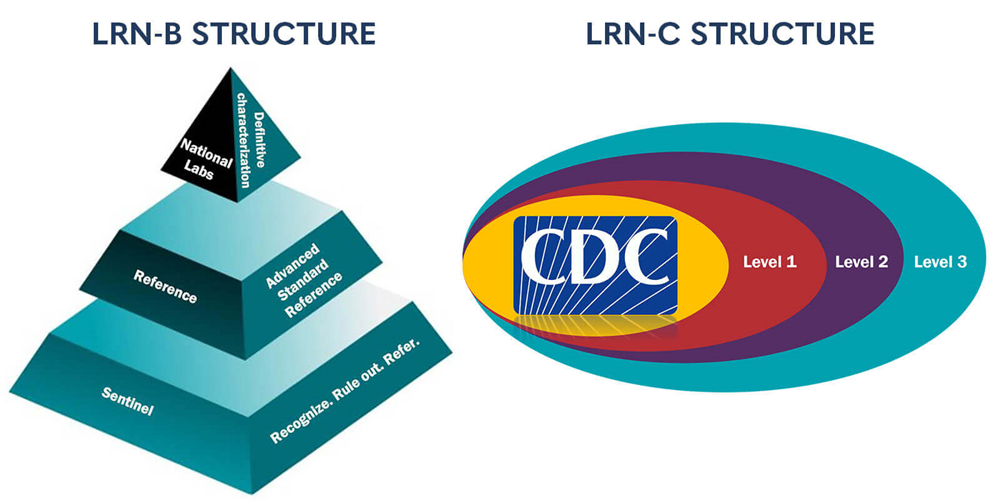
LRN reporting structures
The Laboratory Response Network (LRN) is the nation's laboratory emergency response system for biological, chemical and radiological threats and other public emergencies, such as natural disasters. The LRN is a valuable resource for law enforcement and public health officials during public health emergencies.
Contact the Public Health Preparedness and Response team: [email protected]
Founded in 1999 by APHL, the US Centers for Disease Control and Prevention (CDC) and the Federal Bureau of Investigation to improve US readiness for bioterrorism, the LRN remains a valuable resource for law enforcement and public health officials. It links local, state and federal public health laboratories with sentinel clinical, food, veterinary, environmental and agricultural laboratories, and military and international laboratory centers.
Over time, the US Department of Defense has also become an LRN partner, joining in strategic planning and supporting LRN biological response activities—notably, during the 2014 Ebola outbreak response.
The Laboratory Response Network for Biological Threats (LRN-B) is organized as a tiered pyramid of sentinel clinical, reference and national laboratories. These laboratories are integrated into a single network for rapid detection and response to threats. In 2014, the reference tier was further categorized into reference, standard and advanced laboratories based on capabilities. This configuration:
The Laboratory Response Network for Chemical Threats (LRN-C), the chemical component of the Laboratory Response Network, was established to prepare and respond to chemical threats. The LRN-C links 54 state and local public health laboratories across the US and its territories. These laboratories operate at three levels that reflect their ability to perform various functions during emergency events:

LRN reporting structures
APHL supports the LRN through training, quality improvement initiatives, surge capacity and exercise planning, policy development and fostering of partnerships.
Our activities include:
APHL connects state, local and territorial public health laboratories to promote communication among Bioterrorism Response Coordinators, strengthen their preparedness and response capabilities.
Applications are currently closed.
In the years following the LRN’s founding, significant federal and state investment in public health preparedness strengthened laboratory capability and capacity to respond to health threats, both man-made and naturally occurring. This investment proved invaluable in responding to Hurricane Katrina and Sandy and other natural disasters. It also readied public health laboratories to respond to the nationwide outbreak of 2009 H1N1 influenza and to emerging diseases such as Middle East Respiratory Syndrome (MERS) and Ebola.
To modernize, consolidate and streamline data exchange services, CDC has identified electronic laboratory reporting (ELR) as the desired method for LRN member laboratory reporting to CDC.
ELR is a patient-centric system of reporting that uses a laboratory's existing laboratory information management system (LIMS) to send data to CDC. The system applies to both LRN-B and LRN-C reporting and provides the highest quality data. To date, several LRN-B and LRN-C laboratories have completed the full scope of LRN ELR reporting requirements.
CDC has modernized the data exchange infrastructure for the LRN due to several driving factors, including the need to consolidate and streamline services, the age of the current application used for LRN data exchange and the opportunity to leverage new and more advanced software technologies. After an intensive review of the LRN-B and LRN-C was conducted in 2017, attention was given to the consistent application of vocabulary and messaging standards.
A new standard was finalized in early 2018, which is compatible with electronic laboratory reporting (ELR) (ORU_R01 HL7 v.2.5.1 or ELR to Public Health) requirements consistent with other laboratory data reported to CDC, providing greater data interoperability and flexibility for LRN-B and LRN-C moving forward.
As of March 1, 2025, LRN-B LIMSi-enabled laboratories that have not implemented ELR are required to use DataLink to meet messaging requirements. It will be a requirement of PHEP funding for all LRN-B laboratories and level 1 and 2 LRN-C laboratories to implement ELR for reporting by June 30, 2029.
Contact the LRN Data Integration Team at [email protected] with questions.
The costs for implementing ELR for LRN-B and LRN-C data integration capabilities varies depending on the LIMS platform. There are several funding options available for LRN ELR implementation: