“BEACONS is designed to answer operational questions that only public health laboratories and follow-up programs can address, including data flow, reporting, workforce needs and long-term sustainability.”
APHL Convenes Newborn Whole Genome Sequencing Initiative, Ensuring Public Health Laboratory Input
Can whole genome sequencing (WGS) be integrated into the existing newborn screening system in a way that’s practical, responsible and beneficial for families?
That’s what a new initiative, known as Building Evidence and Collaboration for Genomics in Nationwide Newborn Screening (BEACONS), aims to find out.
BEACONS, funded with a $14.4 million award from the National Institutes of Health, is the nation’s first multi-state, multi-partner genomic newborn screening initiative. APHL is one of several stakeholders taking part in the initiative, coordinating state and territorial participation and offering support with contracting and project management. APHL also manages the initiative’s subawards, facilitates protocol development, supports discussions on data hosting and laboratory information management systems (LIMS) compatibility and helps troubleshoot challenges that arise across states.
“BEACONS is designed to answer operational questions that only public health laboratories and follow-up programs can address, including data flow, reporting, workforce needs and long-term sustainability,” said Sikha Singh, APHL’s director of Newborn Screening and Genetics. “APHL became involved because state and territorial public health laboratories are central to newborn screening, and any future advances must be evaluated within real-world public health environments. As a national convener for the newborn screening community, APHL is positioned to coordinate multi-state work and ensure that public health considerations guide the science.”
Can whole genome sequencing improve newborn screening?
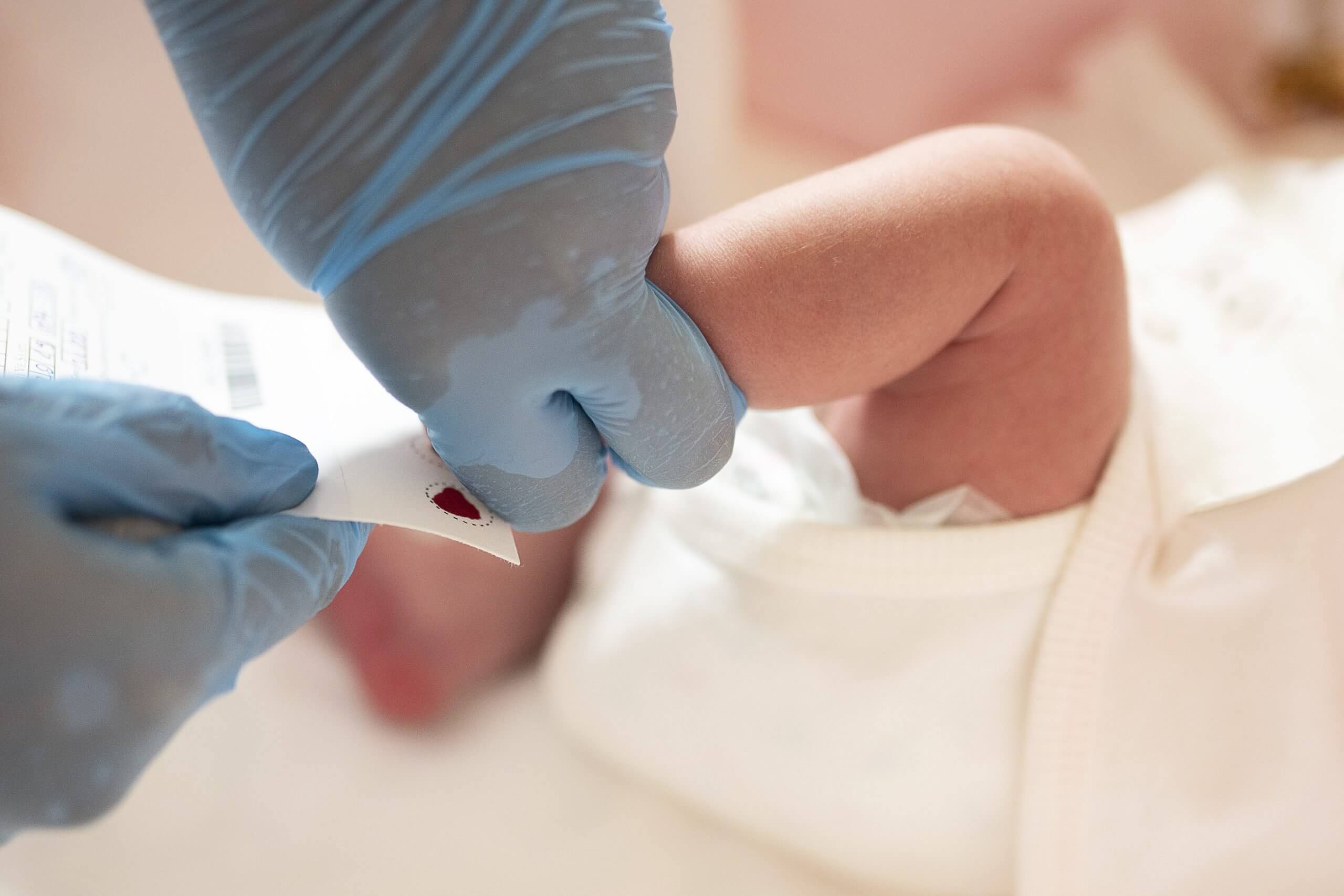
The country’s newborn screening system has been in place for 60 years and is one of the most trusted and successful public health programs in the US. Every year, millions of babies are tested for serious but actionable and treatable genetic conditions, the symptoms of which may not be apparent at birth.
Currently, newborn screening involves taking a few drops of blood from the heel of a baby shortly after birth and sending it to a public health laboratory for analysis. While every state is different in what they test for, the recommended panel includes 38 core conditions (including sickle cell anemia, cystic fibrosis and certain metabolic disorders) as well as congenital heart defects and hearing loss.
Using WGS in the newborn screening program may allow for the detection of disorders not identified through current biochemical methods and/or allow for even earlier disorder identification.
WGS is not meant to replace the existing screening system, Singh emphasized, but act as a layer that complements traditional tests, especially where biochemical markers may not be optimal or where additional genomic detail can improve accuracy.
“BEACONS … allows labs to shape the future of sequencing in newborn screening, rather than having decisions made without public health input.”
“For patients and families, the project may open the door to identifying serious conditions earlier, with clearer genetic information to guide care,” she said. “For some infants, earlier detection could mean faster access to treatment or surveillance. For laboratories, BEACONS gives states a structured space to build genomic capacity and test workflows and learn what is realistic before any large-scale changes are discussed. It allows labs to shape the future of sequencing in newborn screening, rather than having decisions made without public health input.”
Inside the initiative
More than 20 states have expressed interest in taking part in the initiative and the study partners are finalizing a cohort of seven to 10 public health laboratories. In addition to the routine newborn screening all babies receive, parents in participating states and territories will be given the opportunity to have WGS conducted on their baby’s DNA, examining it for hundreds of treatable health conditions. There is no need for another blood draw—blood from the heel prick is also used for the sequencing. When testing is complete, parents will be informed of the results and whether any follow-up care is recommended.
How will the feasibility of using WGS in newborn screening be assessed? Investigators will focus on three specific domains: programmatic, scientific and human. For example, Singh said investigators will look at—among other things—how WGS fits into the routine newborn screening systems, paying particular attention to testing turnaround time, data transfer, cost, workforce capacity and other factors. The study will also examine parent understanding of consent and whether sequencing adds actionable value beyond current screening tests. “If states and territories can perform this work reliably and sustainably at scale, then WGS may be considered feasible for future implementation,” Singh added.
Next steps
The research study will run for approximately three years. At its conclusion, the project team will share its findings with newborn screening programs, federal partners and the wider newborn screening community.
“Any future steps will be guided by the evidence, with a focus on protecting families, supporting public health laboratories and advancing newborn screening in a measured and responsible way.”
“The results will help inform whether larger-scale pilots or implementation studies should follow, and what policies, infrastructure and workforce investments would be required,” Singh said. “Any future steps will be guided by the evidence, with a focus on protecting families, supporting public health laboratories and advancing newborn screening in a measured and responsible way.”