PCR generates amplified DNA that can contaminate other samples, reactions or surfaces, and PCR reactions can be contaminated by other DNA sources. The contamination from previously amplified targets or from patient samples may go unnoticed and could result in incorrect test results including false negatives, leading to the need for extensive lab cleanup and possible temporary lab shut down. Laboratories performing molecular assays should institute procedures to reduce the risk of cross contamination of test samples with amplified DNA products.
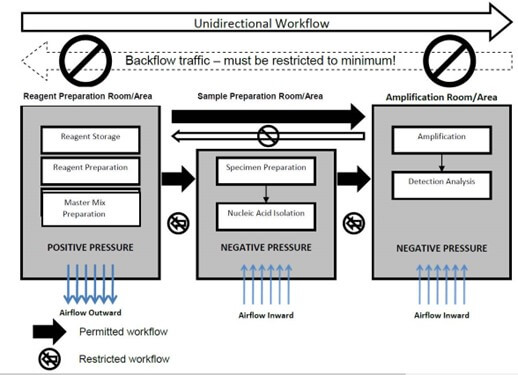
- Unidirectional laboratory workflow
- Operator training and following good laboratory practices
- Use of disposable materials (e.g., gloves, lab coats, consumables)
- Use of DNA-free and nuclease-free water
- Frequent changes of gloves and other PPE
- Placement of a sticky or tacky mat at the entrance of the pre-PCR and reagent rooms
- All staff that enters the laboratory should wear a lab coat appropriate for the room (this includes administrative staff, facilities maintenance, vendors as well as laboratory staff)
- Decontamination of instruments (liquid handlers and centrifuges) after a spill and/or routine cleaning and decontamination, based on laboratory’s standard operating procedure
- Positive displacement pipettes or filter tips for reaction set up
- Aliquoting bulk PCR reagents (e.g., enzyme master mix and water) into small volume single use working stocks
- Separation of dedicated equipment and materials including PPE, for use only in pre-amplification laboratory spaces2
- Inclusion of negative controls, such as no-template controls, water blank controls and normal controls on all PCR plates as indicators of potential contamination.
Inclusion of a no template control on each reaction plate for all assays can help indicate that a contamination event has happened. This reaction has all primers and reagents found in the other reactions on the plate but template DNA is not added. This reaction should ideally show no amplification. Amplification in the no template control indicates a possible contamination, potentially within the assay reagents, during set up of the assay (cross-contamination) or via environmental contamination from aerosolized product within the room.
One method to test for contamination of laboratory surfaces is to perform routine environmental wipe tests. During a wipe test, cotton swabs, or similar, are rubbed along several areas within the lab. Any DNA on the swab is eluted off and the sample is tested for the presence of DNA by running it with the assays used in the laboratory. Lack of amplification indicates that there is no detectable contamination for the amplicon tested.
If a contamination is suspected to be in reagents, these reagents and their derivatives should be thrown out. If a contamination is suspected to be in a work area, replace all disposable supplies and decontaminate the surface areas and equipment. All previously used PPE in the room must be replaced. For reusable supplies, when possible, soak the items in a 10% household bleach solution or DNA-Away® followed by a water rinse; if the item or piece of equipment cannot be submerged, wipe the items thoroughly with a 10% household bleach solution followed by wiping with water.
If an amplicon contamination is found, consider having staff decontaminate the room and/or area that do not work with the assay. This will ensure that the room is not further contaminated during the decontamination process.
Note: A library of all DNA amplicons and their location of use within the laboratory may provide a method to track the source of the contamination.